FDA and 510(k) Assistance
🎯 We are uniquely qualified to assist you in more ways than one:
🚀Assist with US FDA CDRH medical device regulatory compliance through parent/owner company FDASmart including medical device establishment registrations, product classification determinations with relevant CFR regulations that govern, quality systems assessment for cGMP compliance, device design controls and validations, etc.
We have all the necessary tools and experiences WORKING IN YOUR BEHALF.
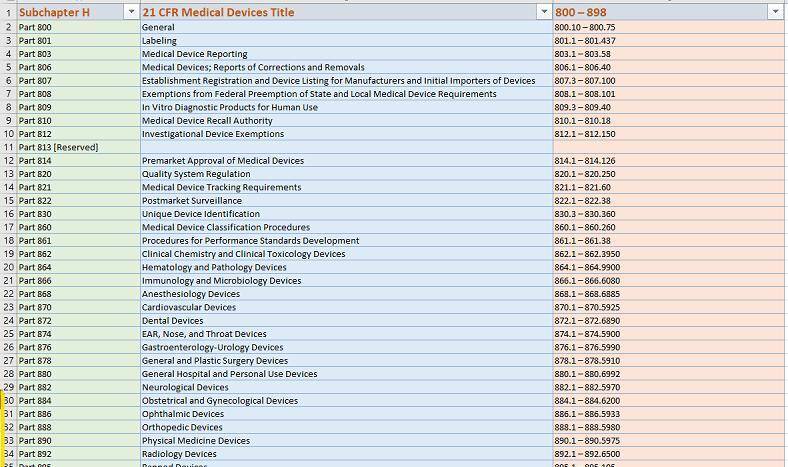
👓As an example, our in-house SmartSearch on Sharepoint cloud, when used for your benefit, allows us to conduct a rich, intelligent search in 21 CFR Part 820 (Quality Systems) without the Internet ‘noise’ that betters US FDA website own search engines.
The same applies to ALL Other 21 CFRs on medical devices (US FDA Subchater ‘H” – 34 CFRs in all)
See sample 21 CFR Part 820 Search for any statute referencing ‘quality’ ‘supplier’ as shown below:
(* Challenge: NOW – Try to search for “quality” “supplier” at FDA website for BOTH KEYWORDS referenced by the same statute *)
https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?CFRPart=820&showFR=1
👓 For IVD (In Vitro) test developers, as another example, we can SmartSearch all 73+ CDRH Virtual Town Hall recorded transcripts IN ONE PLACE to prepare an intelligent industry review and survey for your medical device development during this window of EUA opportunity !
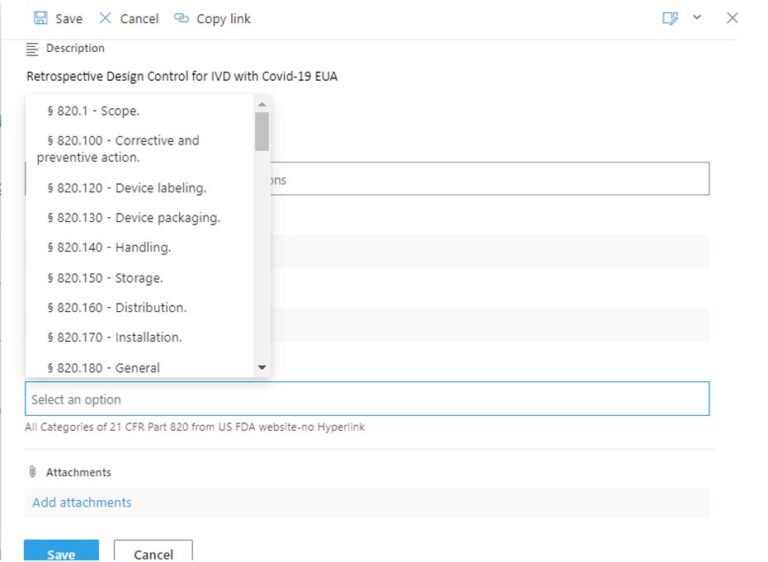
👓 Yet another example for early-stage medical device companies that benefited with US FDA on its waiver of most 21 CFR 820 regulation (Quality Systems) in order to accelerate EUA submissions whether IVD or nIVD medical devices, you know have the issue of RETROSPECTIVE COMPLIANCE for your 21 CFR 820 quality systems that include design control life cycle, validation, risk Management, etc.
We can assist with this by deploying Microsoft’s out-of-the box Task project management app further enhanced by eSTARHelper customization on the task/subtask data entry by forcing to include and anchor each task against a specific 21 CFR 820 predicate rule.
See sample screen for your retrospective Quality Systems regulatory task management, i.e each task anchored against a specific 21 CFR 820 rule for US FDA regulatory proof of concept and compliance.
🚀Assist with IT/Cloud solutions unsing the Microsoft Cloud platform including Azure, Office 356, Sharepoint Online and Teams.
For IVD (In Vitro) test developers, as another example, we can SmartSearch all 73+ CDRH Virtual Town Hall recorded transcripts IN ONE PLACE to prepare an intelligent industry review and survey for your medical device development during this window of EUA opportunity !
🎯 We realize there’s no one-size-fits-all for medical device companies!
So today’s eSTARHelper (Version 1.0) serves as an added-value benefit in two ways:
- Accelerates the installation of eSTARHelper OOTB/Customized features on Microsoft Teams/Sharepoint already built into your own cloud tenant site to cut development costs
But also…
- Allows for your own environment customizations. Most of today’s legacy or re-purposed haphardly into cloud-based QMS are fairly static, and forces your enterprise to adopt what their platforms offer. Not so with eSTARHelper hosted in Microsoft’s Office 365/Sharepoint and Teams platforms.
Medical device companies come in all types (IVD-InVitro or nIVD-Not Invitro, etc), can be big or small, locally centralized or mostly remote work forces employed and so on.
You maybe doing your very first 510K Premarket Submission or making modfications to an existing one of hundred 510Ks already authorized with substantial equivalence to an existing US medical device product.
You maybe one of the major global medical player or a small start-up seizing the newfound market opportunities with US medical devices under an EUA (Emergency Use Authorization).
At the onset of Covid-19, FDA CDRH has opened up the Emegency Use floodgates to ease and waive certain customary regulations but, bear in mind, FDA, can at its own sole discretion, withdraw any EUA to expire when it deems appropriate for the American healthcare industry, e.g. a Covid-19 detection kit manufacturer can begin to sell its products under an EUA only to be marginalized after EUA expiration if caught without a 510K already in places or at least in motion.
US FDA is hard at work in crafting a transition pathway from EUA to 510K with an official Guidance but it’s not been released. The time to evaluate 510K premarket submission is NOW not then the plug has been pulled so to speak.
That aside—the regulatory terrain for medical devices in Unites State markets will remain challenging to navigate on your own.
👀 We can assist YOU in more ways than one at NO COST for the initial consultation or needs assessment
👍 As a Microsoft Clourd Partner, we can assist to assess your Microsoft Office 365/Sharepoint or Microsoft Teams cloud options to host eSTAR online in a secure, encrypted and fully authenticated private tenant site
👍 As concept & actual app developers of eSTAR hosted in Microsoft Teams Cloud environment, assist with the best fit Teams site map and options available for your medical device company.
For IVD (In Vitro) test developers, as another example, we can SmartSearch all 73+ CDRH Virtual Town Hall recorded transcripts IN ONE PLACE to prepare an intelligent industry review and survey for your medical device development during this window of EUA opportunity !
👍 Free Initial compliance assessment with 21 CFR 820 and may other CFR quality systems obligations mandated by FDA CDRH
👍 Free initial assessment of your FDA regulatory compliance with CFR statutes on medical devices
👍 Extrapolating just what a US FDA Guidance means in terms of FDA’s current thinking (* subject and open to wide interpretation for fulfillment*)
👍 Introducing eSTAR PDF template and then some behind-the hood tricks, e.g. go beyond eSTAR help screens, learn how to meet 21 CFR Part 11 for electronic documents with eSTAR
👍 Assist with US FDA establishment registrations, MDUFA fees request for Small Business classification and discounted fees
👍 Determine your medical device product classification and CFR regulatory requirements for full regulatory compliance